Oxalic Acid H2c2o4*2H2O Ethanedioic Acid Oxalic Acid - China Oxalic Acid, Oxalic Acid 99.6% Min | Made-in-China.com

Details of structurally characterized anhydrous and hydrated oxalic... | Download Scientific Diagram
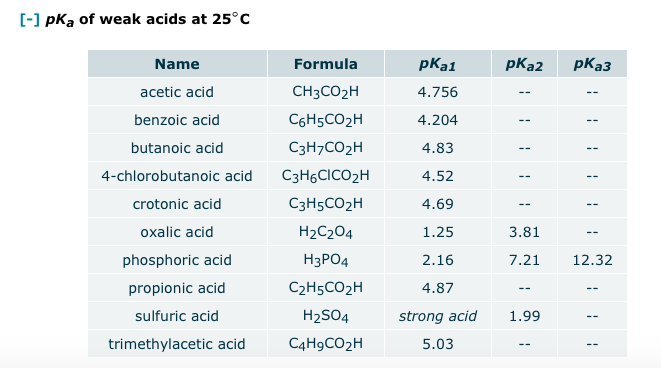
![SOLVED: Suppose an aqueous solution of oxalic acid (H2C2O4) is prepared. Calculate the equilibrium molarity of C2O4^-2. Round your answer to significant digits. [-]pKa of weak acids at 25°C Name Formula pKa1 SOLVED: Suppose an aqueous solution of oxalic acid (H2C2O4) is prepared. Calculate the equilibrium molarity of C2O4^-2. Round your answer to significant digits. [-]pKa of weak acids at 25°C Name Formula pKa1](https://cdn.numerade.com/ask_images/a8572c9feb7a4721935281facc93d84d.jpg)
SOLVED: Suppose an aqueous solution of oxalic acid (H2C2O4) is prepared. Calculate the equilibrium molarity of C2O4^-2. Round your answer to significant digits. [-]pKa of weak acids at 25°C Name Formula pKa1


![Oxalic Acid [HOOCCOOH] Molecular Weight Calculation - Laboratory Notes Oxalic Acid [HOOCCOOH] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/oxalic-acid-molecular-weight-calculation.jpg)













![Oxalic Acid Dihydrate [HOOCCOOH.2H2O] Molecular Weight Calculation - Laboratory Notes Oxalic Acid Dihydrate [HOOCCOOH.2H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/oxalic-acid-dihydrate-molecular-weight-calculation.jpg)