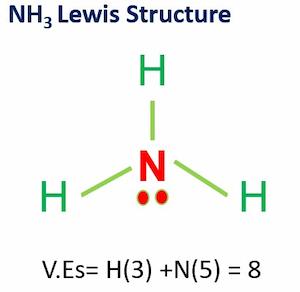
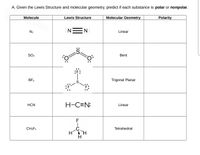
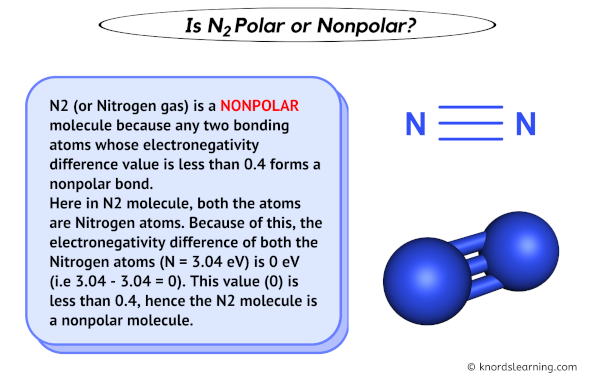
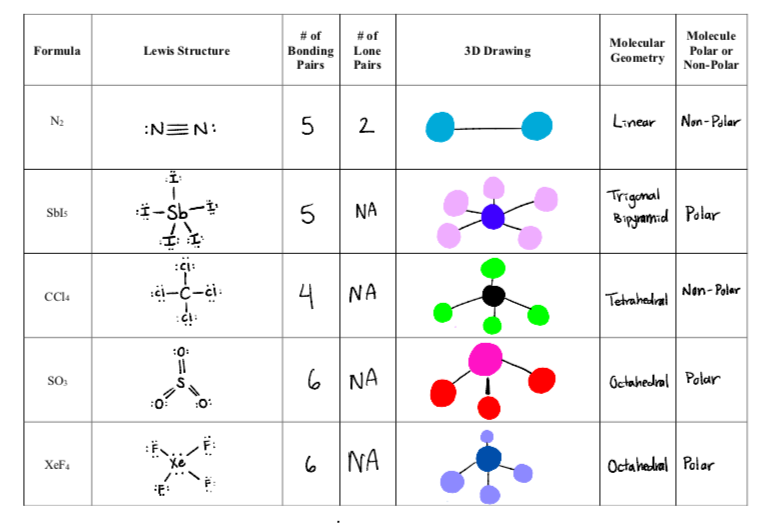
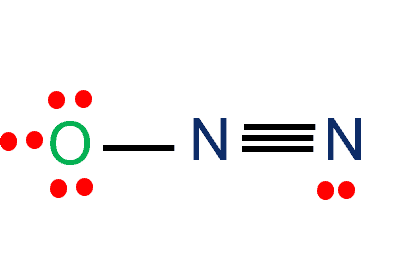
Draw the Lewis structure for N2 and state its molecular geometry. Is it polar or nonpolar? | Homework.Study.com

Is N2 Polar or Non-polar? (Nitrogen Gas) | Is N2 Polar or Non-polar? ( Nitrogen Gas) N2 is a chemical formula for Nitrogen gas and in today's video, we help you determine if
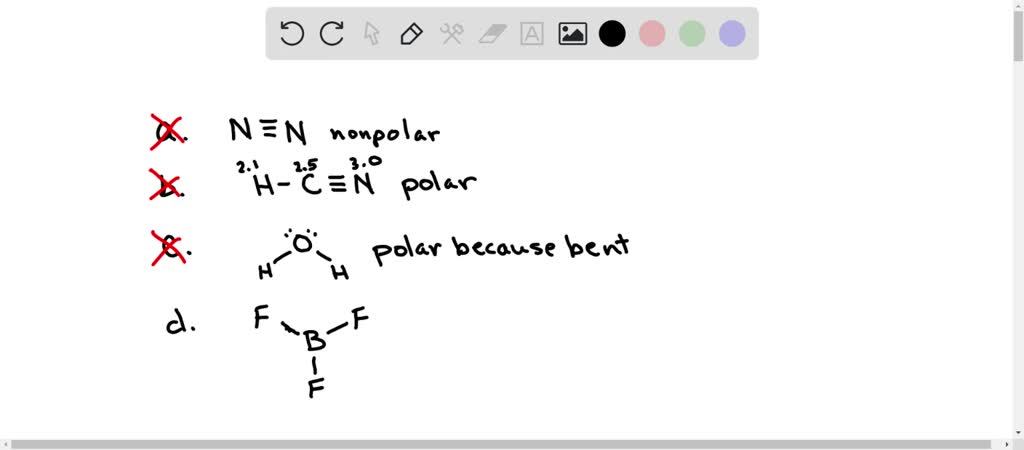
SOLVED: Which of the following statements concerning molecular polarity is correct? Select one: a. All diatomic molecules are polar: H2, N2, HF, and Cl2. b. All linear triatomic molecules are non-polar: CO2,