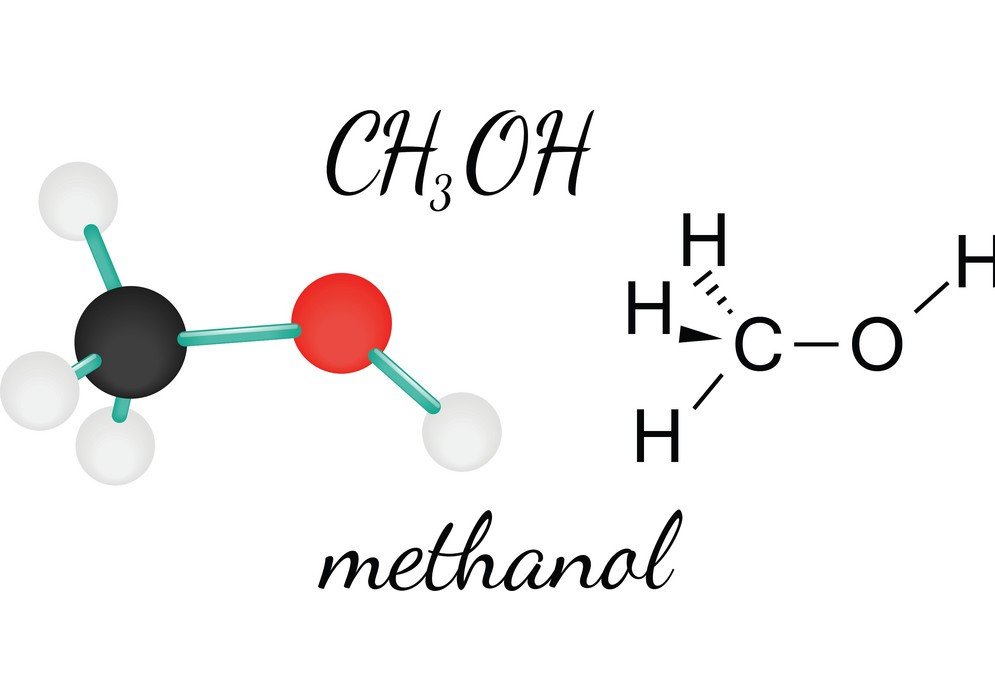
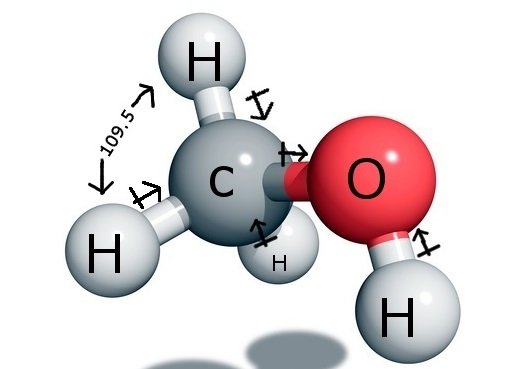
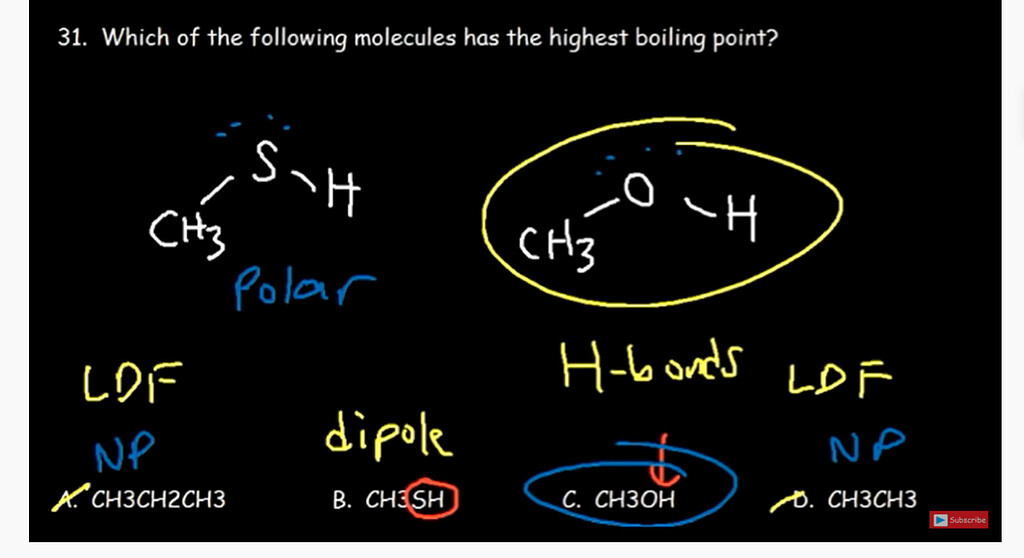
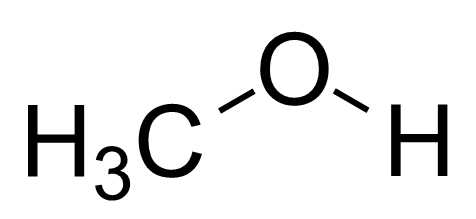
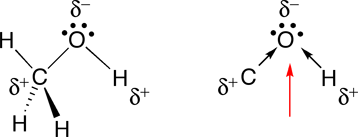SOLVED: 5. (5 pts) Classify each of the following as polar (P), nonpolar (NP), ionic (I), or metallic (M). CO2 CH3OH NH3 PCl5 SiCl4 NaCl CHCl3 COH2 (methanol) Fe

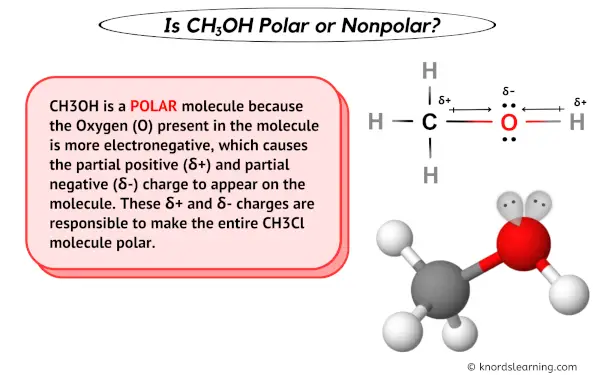
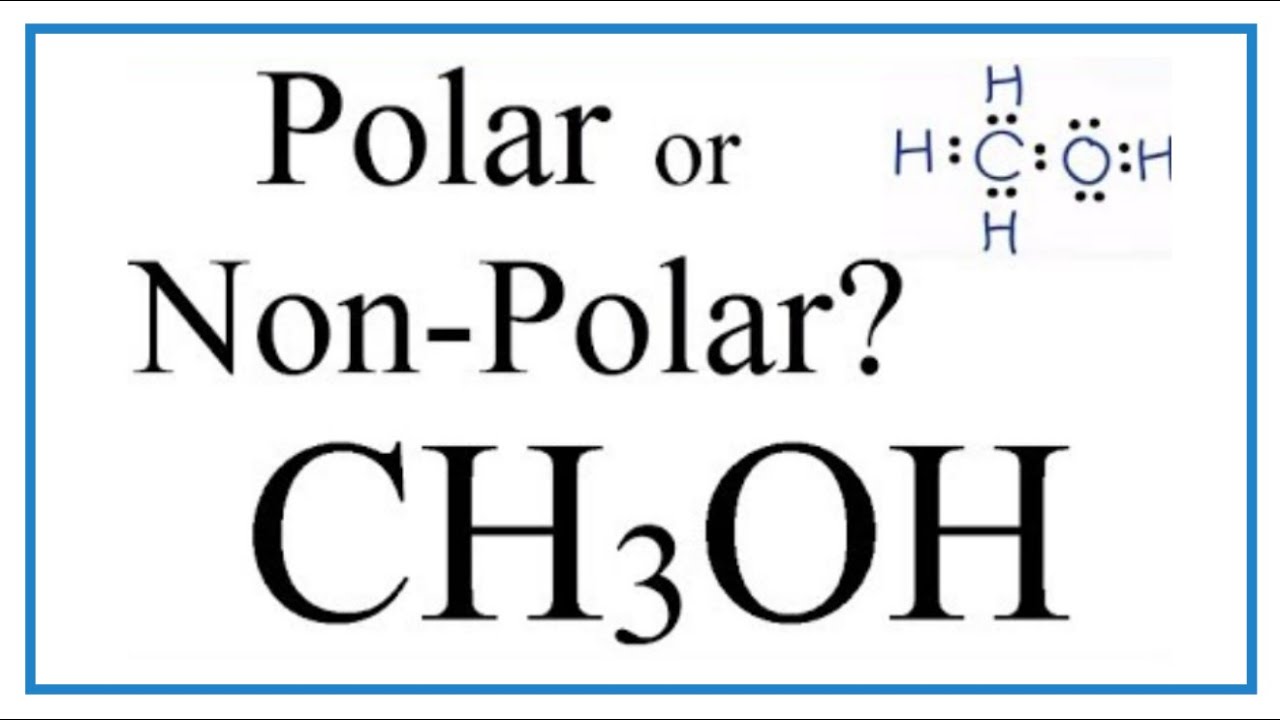
Is CH3OH Polar or Nonpolar? - Polarity of Methanol | Polar, Covalent bonding, Electron configuration
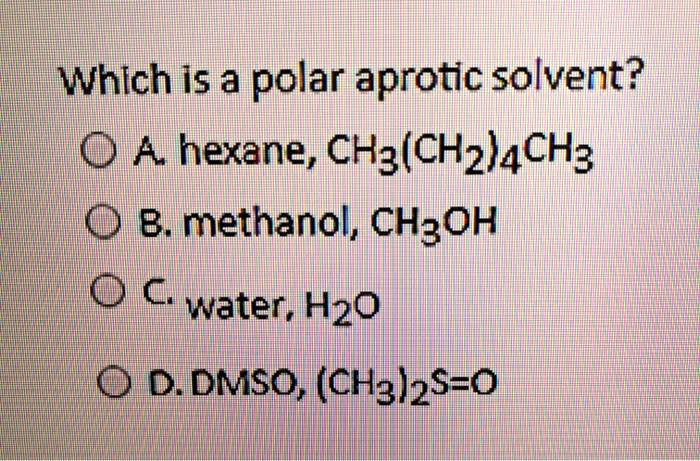
SOLVED: Which is a polar aprotic solvent? A. hexane, CH3(CH2)4CH3 B. methanol, CH3OH C. water, H2O D. DMSO, (CH3)2S=O