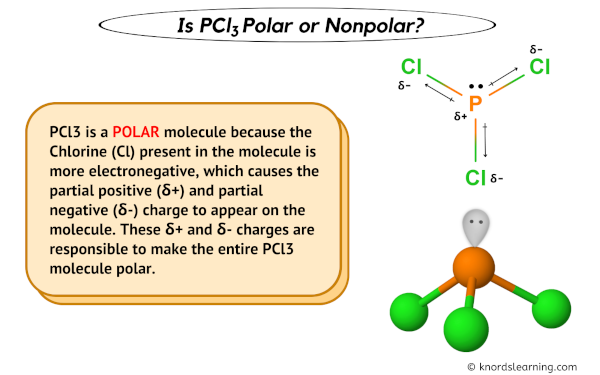
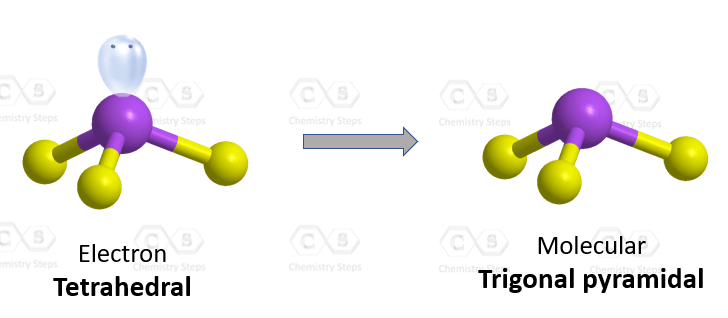
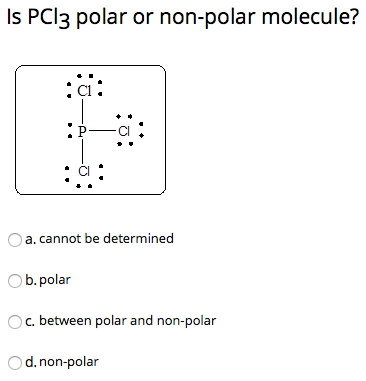
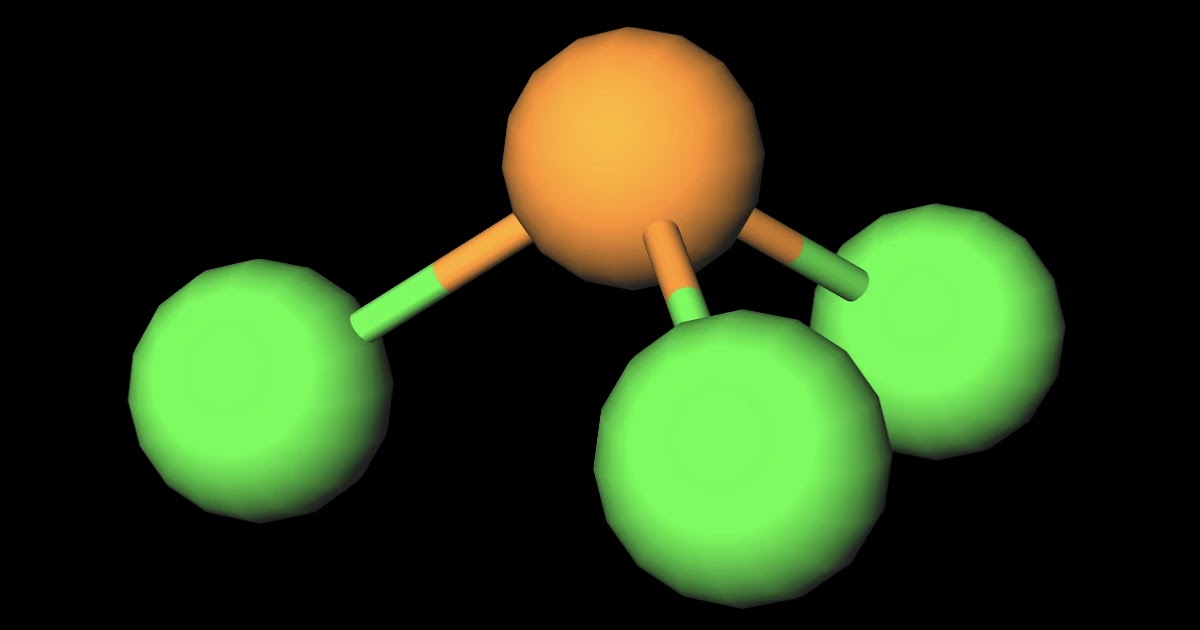
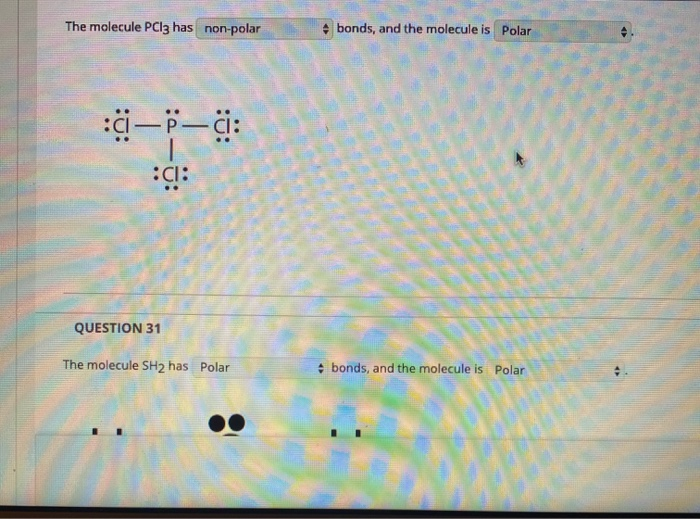
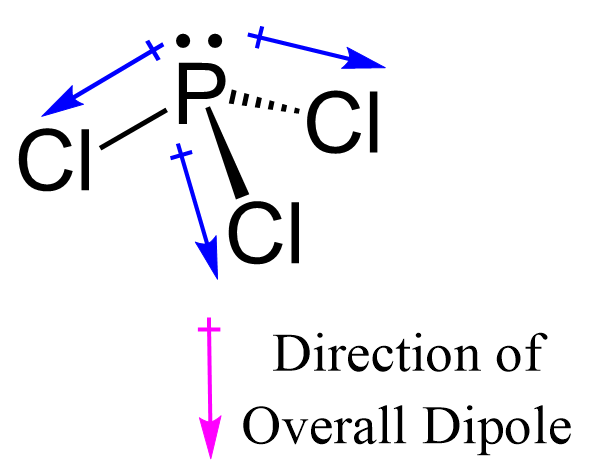
Which of the following are characteristics of phosphorus trichloride, PCl3? Select all that apply. a. trigonal planar b. one unshared pair of electrons on P c. sp2 hybridized at P d. polar

Is PCl5 Polar or Nonpolar? (Phosphorous Pentachloride) | Is PCl5 Polar or Nonpolar? (Phosphorous Pentachloride) PCl5 is the chemical formula for Phosphorus Pentachloride. Phosphorus atom in the center forming... | By Geometry
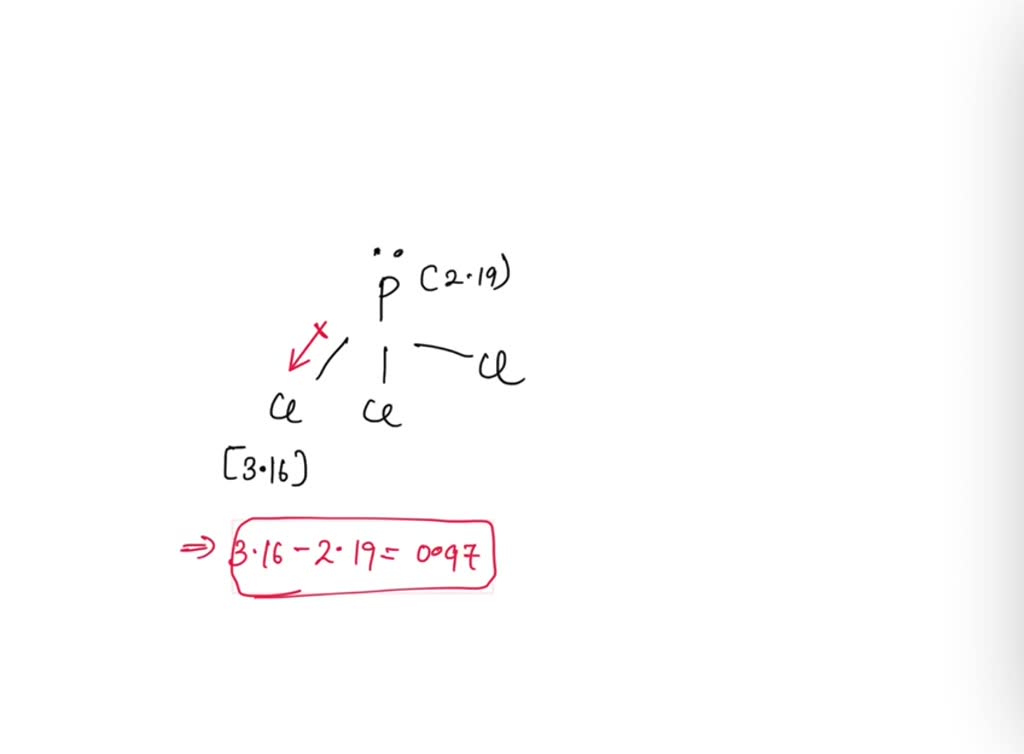
SOLVED: Show how to calculate the difference in electronegativity for the bonds in phosphorus trichloride, PCl3, then classify the bonds as being nonpolar covalent, polar covalent or ionic